The global Vascular Closure Devices Market is projected to reach USD 1.3 billion by 2026, at a CAGR of 7.0% during the forecast period. The growth in this market can be attributed to the large number of approvals for vascular closure devices, high prevalence of cardiovascular diseases, increasing prevalence of obesity, increasing focus of market players on large-bore vascular closure devices, growing geriatric population, favorable reimbursement scenario for interventional radiology procedures, and rising focus on effective blood loss management in patients during surgeries. Emerging markets providing growth opportunities for market players and growth in the number of hospitals & surgical centers are expected to offer strong growth opportunities for players in the market. In contrast, the need for highly skilled professionals, risks associated with vascular closure devices and stringent regulatory framework may challenge market growth to a certain extent. The vascular closure devices market is segmented based on type, access, procedure, and region.
Download a FREE Sample Report PDF of the Global Vascular Closure Devices Market Research Report at https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=180101468&utm_source=Ganesh&utm_medium=P
Vascular Closure Devices Market Key Players
The prominent players operating in this market include Terumo Corporation (Japan), Abbott (US), Cardinal Health (US), Cardiva Medical, Inc. (US), Medtronic Plc (Ireland), Morris Innovative, Inc. (US), Teleflex Incorporated (US), Merit Medical Systems, Inc. (US), Vasorum Ltd. (Ireland), TZ Medical, Inc. (US), Vivasure Medical Ltd. (Ireland), Tricol Biomedical (US), Scion Biomedical (US), Advanced Vascular Dynamics (US), Marine Polymer Technologies, Inc. (US), Transluminal Technologies (US), Rex Medical (US), Meril Life Sciences Pvt. Ltd. (India), Medas USA (US), and Vygon Company (France).
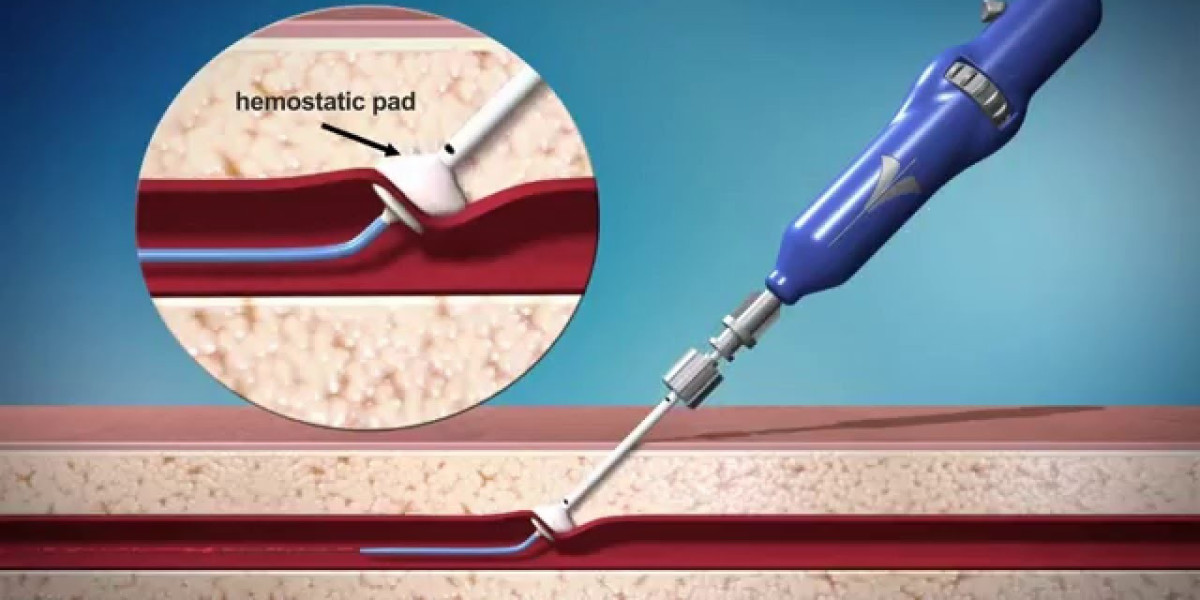
Passive approximators segment dominated the Vascular closure devices market in 2020.
Based on type, the vascular closure devices market is segmented into passive approximators, active approximators, and external hemostatic devices. Passive approximators account for the largest share of the vascular closure devices market. The large share of this segment can be attributed to the increasing number of approvals for vascular closure devices and the wide range of advantages associated with these devices, such as ease of use and reduced complication rate in patients.
The interventional radiology segment to witness the highest CAGR during the forecast period.
Based on procedure, the vascular closure devices market is segmented into interventional cardiology procedures and interventional radiology/vascular surgery. Interventional cardiology for the largest share of the vascular closure devices market. The large share is attributed to the diagnosis and treatment of numerous cardiovascular disorders due to advancements in angioplasty and cardiac stenting and the growing prevalence of cardiac disorders globally.
North America was the largest regional market for vascular closure devices market in 2020.
The global vascular closure devices market is segmented into North America (the US and Canada), Europe (E-3, and the Rest of Europe), Asia Pacific, and Rest of World. In 2020, North America dominated the global vascular closure devices market, followed by Europe. The large share of the North American market can be attributed to the rising prevalence of cardiovascular diseases, the growing trend of one-day surgeries for vascular procedures, increasing research and clinical trials for vascular closure devices in the US, the increasing number of cardiac arrest cases, and the long waiting periods for cardiac surgeries (which encourage the adoption of minimally invasive procedures) in Canada.
This report categorizes the vascular closure devices market into the following segments and subsegments:
By Type
- Passive Approximators
- Collagen Plugs
- Sealant or Gel-based Devices
- Compression Assist Devices
- Active Approximators
- Suture-based Devices
- Clip-based Devices
- External Hemostatic Devices
By Access
- Femoral Access
- Radial Access
By Procedure
- Interventional Cardiology
- Interventional Radiology/Vascular Surgery
By Region
- North America
- US
- Canada
- Europe
- E-3
- Rest of Europe
- Asia Pacific
- Rest of World
Direct Purchase of the Global Vascular Closure Devices Market Research Report at https://www.marketsandmarkets.com/Purchase/purchase_reportNew.asp?id=180101468
Recent Developments:
- In 2021, Vivasure Medical Ltd. (Ireland) announced the development of its product, PerQseal Blue. This product is a large-bore vascular closure device used after interventional cardiovascular procedures.
- In 2021, Abbott (US) acquired Walk Vascular, LLC (US) to expand its portfolio of peripheral vascular devices.
- In 2020, Teleflex Incorporated (US) acquired Z-Medica, LLC (US), an industry-leading manufacturer of hemostatic products that will lead to revenue growth for the company by expanding its product portfolio in the Interventional segment.
- In 2018, Cardinal Health (US) launched MYNX Control, a sealant or gel-based device with extravascular sealing and resorbability properties.